Dutch Company Will Start Selling Gene Therapy Treatment in 2013
November 6, 2012
A Dutch company, uniQure, states that it will start selling the first human gene therapy to be approved in the West by mid-2013. They expect an explosion of similar therapies to be available soon.
Glybera was approved by the European Commission on October 25. This drug treats the extremely rare disorder lipoprotein lipase deficiency (LPLD) and it’s the first to be approved for sale in Europe or North America.
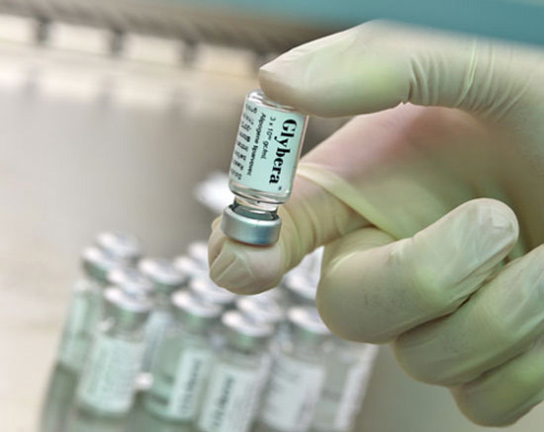
Glybera gene therapy drug.
Gene therapy modifies a patient’s DNA to combat specific diseases, and has been used experimentally to treat blindness, depression, and brain wasting diseases.
These treatments have struggled to obtain regulatory approval in the West. In China, gene therapy has been used for treating head and neck cancer as far back as 2003.
Glybera treats LPLD, a disease which is inherited, and affects about one or two people in a million, preventing them from metabolizing certain fat particles in the blood. Glybera will cost insurance companies $1.6 million per dose.
The company is developing other gene therapies to treat diseases including blood clotting disorder haemophilia B, metabolic disorder acute intermittent porphyria, central nervous system disorder Parkinson’s and enzyme disorder Sanfilippo B.
No comments:
Post a Comment